Verily Lifesciences received the approval for their new Verily Patch product and the associated app.
Verily Patch is the wearable vital signs monitor that is being offered by the company. This approval comes on a day when the company announced a new $700 capital investment.
Related reading
- Verily researching a new finger wearable continuous blood pressure monitor
- Verily Life sciences announce results of Baseline Sleep Mission Study
- Google’s Verily Sets Up JV to help Millions of Sleep Apnea Sufferers
- Best free exercise apps to strengthen and help relieve pain
- Biointellisense gets regulatory clearance for its BioButton wearable
Verily’s new wearable vital signs monitor development.
Verily today announced a $700 million investment round from current investors, including Alphabet, Silver Lake, Temasek, Ontario Teachers’ Pension Plan, and others. The capital will be used to support the rapid expansion of some of Verily’s leading commercial businesses.
The request for approval was filed on Oct 5th, 2020, and was recently approved.

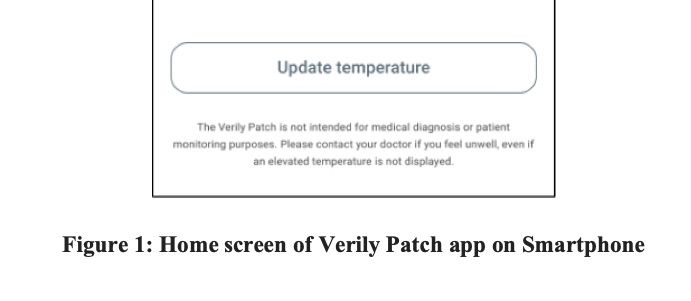
From the various screenshots embedded in the filing, it appears that the patch helps with remote monitoring of vital signs.
According to the filing, the new Verily Patch is not intended for medical diagnosis or patient monitoring purposes. Does it mean that the new wearable patch sensor is something that Verily plans on using for internal studies or more as a wellness monitor?
The company already has a wearable product called the Study Watch, which collects biometric health information, such as ECG, heart rate, electrodermal activity, and inertial movements.
Data from the study watch is processed and stored in their cloud platform using proprietary algorithms and translated into program-specific dashboards or insights.
It is unclear from the reg filing how the new wearable vital signs patch will be used.