FDA has cleared the oxehealth Vital Signs platform as a part of a new DeNovo filing that was spotted today. The software provides an optical camera-based contactless measurement of pulse, heart, respiratory, and more.
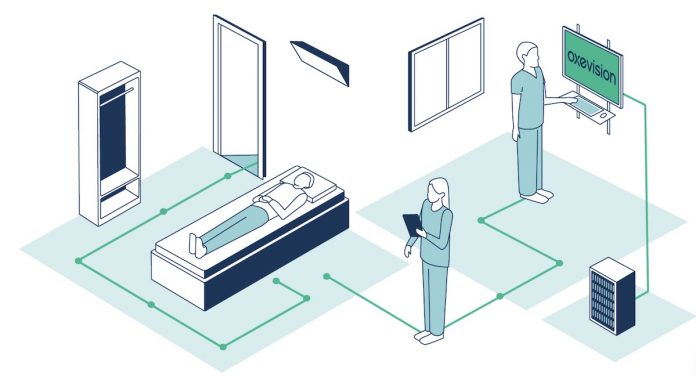
Made by a UK-based startup with solid ties to NHS and the European health community, Oxehealth Limited, the remote monitoring platform is unique because it does not require a wearable sensor to be used on the patient’s body.
Instead of computer vision, the platform uses cameras and sensors to detect and read essential parameters such as pulse and respiratory rate.
Contents
Related Reading:
- How to add your vision prescription to your iPhone’s Apple Health app
- Google Plan of a Future Bathroom Includes Automatic Health Monitoring
- Apple is developing Apple-branded health hardware products
- Ava receives FDA clearance for its ovulation tracking bracelet
- This FDA-Approved Gadget Treats Migraines Without Medicine
How does Oxehealth monitoring platform work
When your heart beats, your skin flushes red. The human eye cannot see these “micro blushes,” but Oxehealth’s optical sensor can, using infrared light and our proprietary signal processing techniques. Our software counts the micro blushes to calculate a pulse rate.
The scientific breakthrough is the platform’s ability to use PPG to detect pulse rates from several meters away.
Oxehealth’s system calculates breathing rate by counting the movements of the body created by chest and diaphragm breathing, just as a nurse would if they were observing the patient with a stopwatch.
The system is similar to a feature unveiled in the latest Google’s Nest hub. The ‘Sleep Sensing’ Google feature allows for sleep tracking via a motion detector.
Additional Patient monitoring features
Oxehealth’s Vital Signs platform can track the patient’s vitals and provide additional motion-based monitoring services such as sleep monitoring, overnight activity monitoring, and out-of-room alerts.
The platform consists of a secure optical sensor installed in a patient’s room. The sensor continuously tracks and obtains contact-free readings from the patient, no matter where they are in the room. Information and alerts are automatically provided to health care facilities.
The company’s Vital Signs module is a class IIa medical device cleared in Europe. It is not available for sale, demonstration, or trial use in any country outside the EU or UK.
With the latest DeNovo clearance, this contactless monitoring platform could soon find its way into US health facilities.
Oxehealth secured financial commitments totaling £10m this month, which will be delivered in two rounds across 2021 and 2022. The rounds have been underwritten by existing shareholders and will be used to fund the commercial expansion of Oxevision, a vision-based patient monitoring, and management platform.