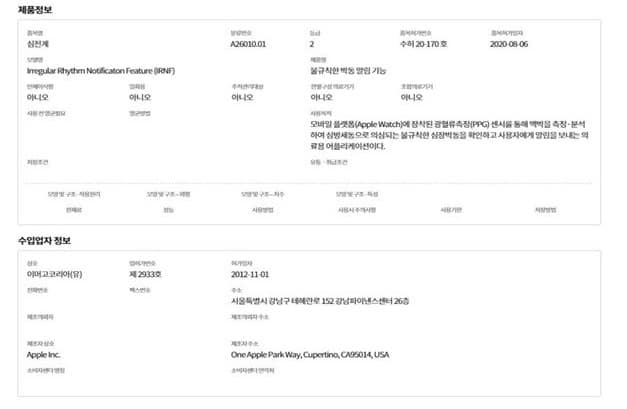
Apple obtained approval from the Ministry of Food and Drug Safety (MFDS) in South Korea regarding its electrocardiogram (ECG) application.
Related:
- Apple’s new ECG feature for detecting AFib at high HR gets FDA Clearance
- Fitbit finally wins regulatory clearance for Afib (Atrial Fibrillation) in the US and Europe
- Samsung Galaxy Watch 3 Health features Vs Apple Watch Health Detailed
- Apple’s AI-based odor sensor technology could transform future Apple Watch Health and Safety offerings
- The New Oppo Watch is real nice but Apple has little to worry
ECG comes to Apple Watch in Korea…finally!
Users in South Korea will now be able to use the ECG feature on their Apple Watch.
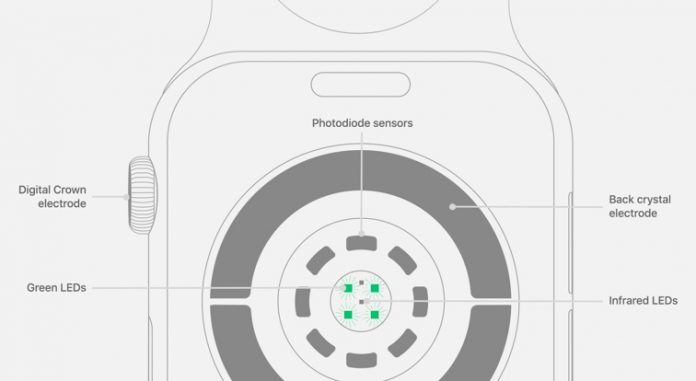
Apple’s “irregular heartbeat alarm feature” measures and analyzes one’s pulse through a photoplethysmography (PPG) sensor that is installed in Apple Watch.
It looks for a sign of an irregular heartbeat that is suspected of atrial fibrillation and alerts users.
According to etnews, where this story was originally revealed, it is expected that Apple Watch’s ECG application will also be widely used in South Korea as Apple obtained approval from the MFDS.
Apple will be able to provide the service after a simple update to the application since it already completed every prerequisite including adding a Korean version to the application.
The ECG app and irregular heart rhythm detection notification feature were released in the US way back in 2018.
Originally, the feature was only available on Apple Watch Series 4 in the US, Puerto Rico, Guam, and US Virgin Islands when it was released.
Here are the countries where the aFib notification feature is currently available.
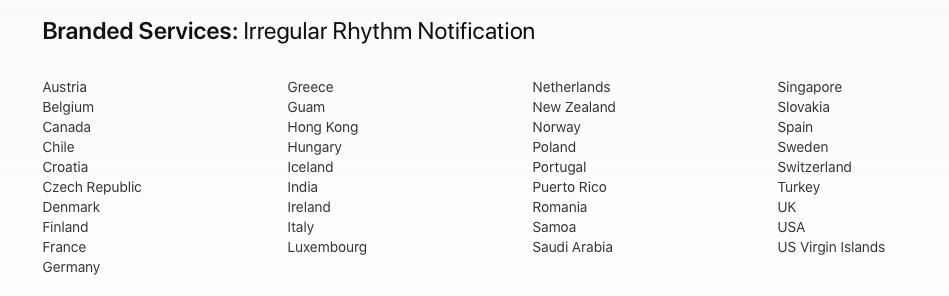
It is surprising that it is taking this long for Apple to obtain approvals in other countries and roll out the feature in different geographies.
Apple is not unique when it comes to MFDS delays in South Korea.
The ECG sensor on the Galaxy Watch Active 2 has gone unused for the better part of a year. Samsung received approval to use the ECG feature for measuring heart rhythms from South Korea’s Ministry of Food and Drug Safety in May 2020.