Pear Therapeutics is emerging as a leader in the area of digital therapeutics. The company was the first to obtain FDA approval for a novel digital prescription therapeutic designed to help patients suffering from substance use disorder.
Last year, Pear Therapeutics obtained FDA approval for Somryst, it is a prescription digital therapeutics for treating chronic Insomnia.
Related Reading:
- Fitbit Health Solutions details out numerous exciting in-flight health initiatives
- Fitbit teams up with NASA offering health and safety services to astronauts and employees
- Fitbit Introduces Blood Glucose integration and more in its latest update
Now the company is collaborating with Mayo Clinic on a new clinical trial of hundred patients to assess the real-world effectiveness of a mobile-delivered, digital therapeutic device delivering cognitive behavioral therapy for Insomnia.
This new study will use a novel patient-centered data-sharing platform with linkage to Fitbit. The new platform with Fitbit integration is called “Hugo.”
Half of the cohort with insomnia will receive the Somryst digital therapeutic link to the Hugo platform and Fitbit (Inspire 2). Half of the patients with insomnia will not receive the Somryst. Still, they will receive a Fitbit and be enrolled in the Hugo platform. ![]()
The treatment duration will be 9 weeks with a 21-, 35-, and 61-week follow-up.
The Somryst intervention will deliver CBT-I via mobile devices as 6 treatment core modules over 9 weeks.
Additionally, the Hugo platform will collect patient-generated engagement data, healthcare utilization outcomes, and patient activity/clinical outcomes.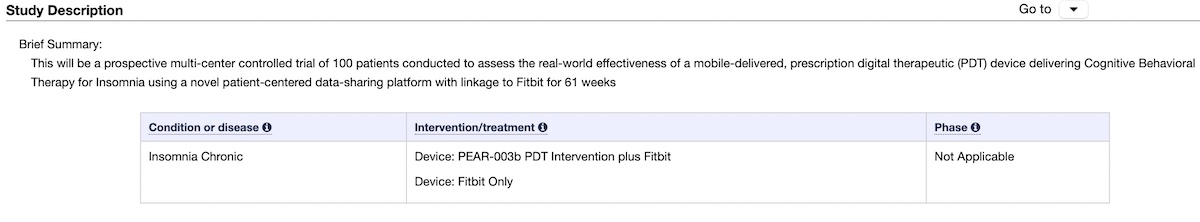
Yale University is sponsoring the new study.
The new clinical trial was announced this week and marked another win for wearable integration in digital health and therapeutics.
As more and more folks start using wearables such as the Fitbit and Apple Watch regularly, digital health companies such as Pear Therapeutics explore ways to link their product offerings with these wearables.
According to the American Sleep Association: Nearly 70 million Americans have a sleep disorder. Insomnia is the most common sleep disorder, with 30% of adults experiencing short-term insomnia. About 10% of people have long-lasting insomnia.
Fitbit has about 30 million users with only half a million paying subscribers on its premium platform.
Fitbit currently offers sleep monitoring features on its platform. The company’s new software update can also detect snoring and assign a sleep profile based on a user’s sleep patterns.
Integrations such as these, if available, will help strengthen Fitbit’s premium offerings in the long term and will attract new users who are looking to proactively manage their health beyond measuring the number of daily steps.







